Enforce and monitor compliance in automated market research workflows.
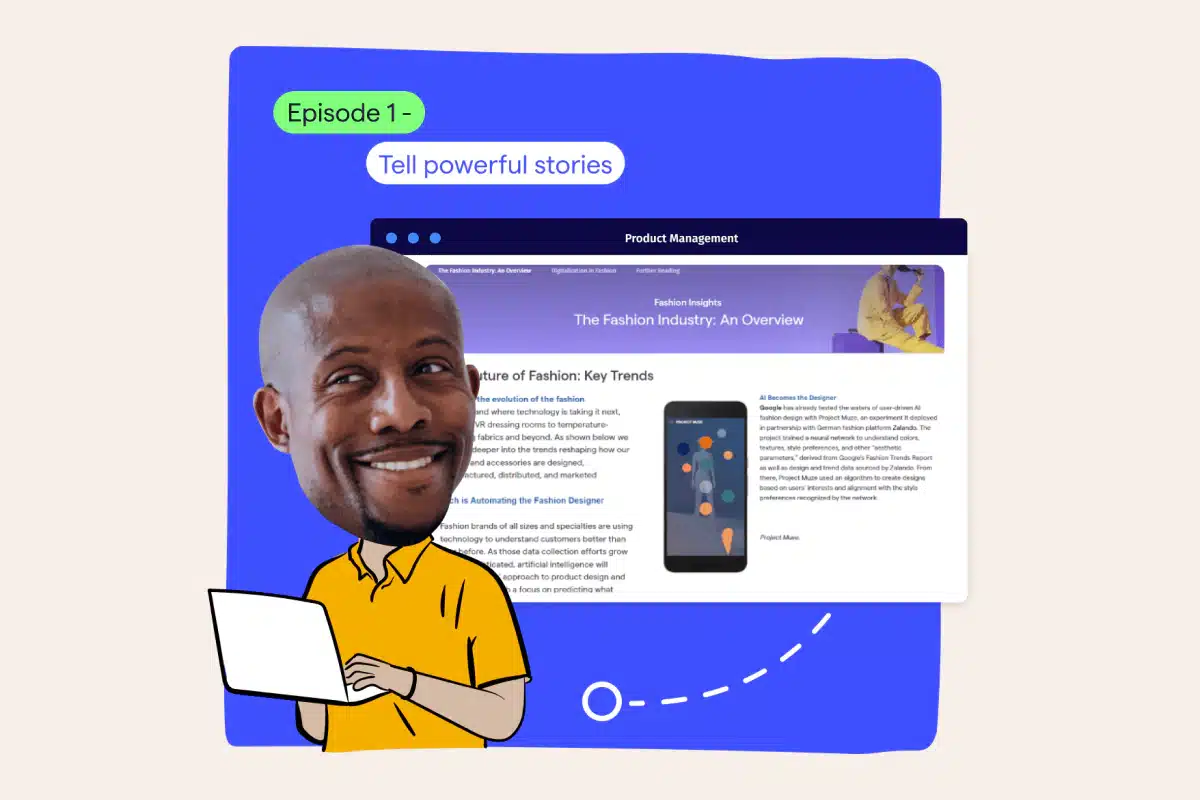
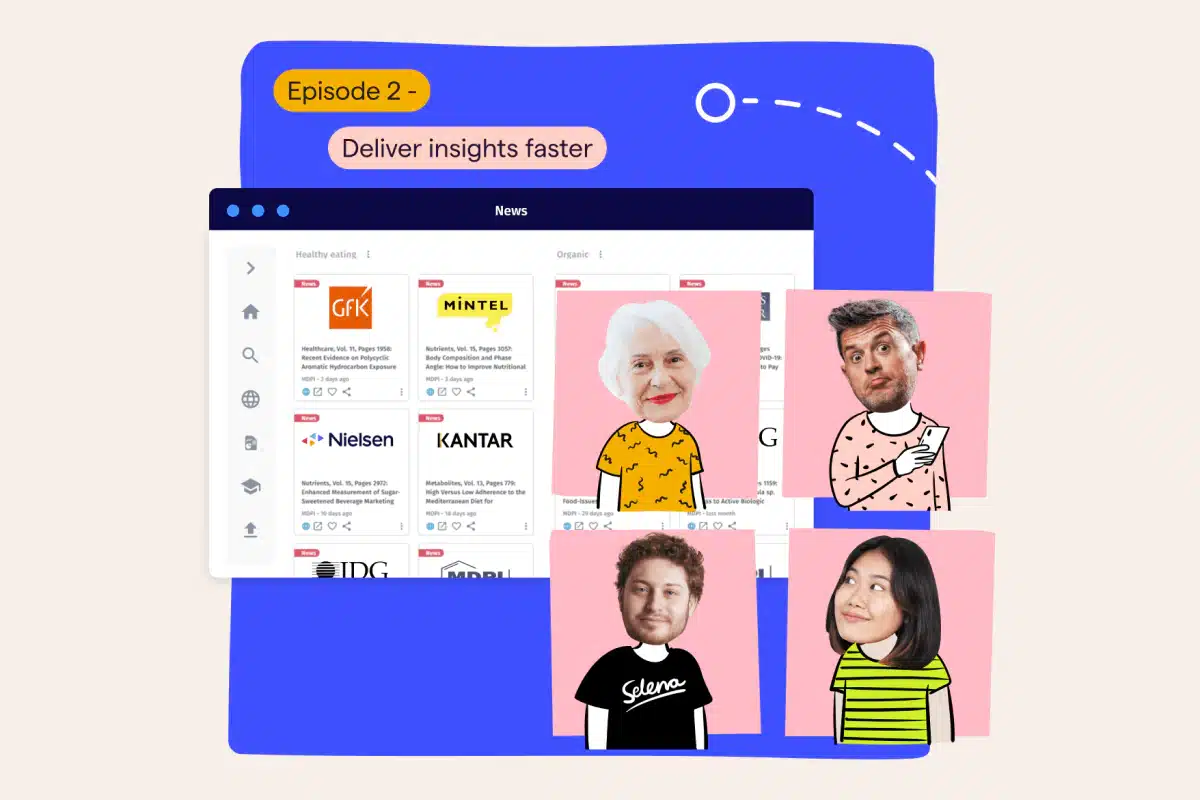
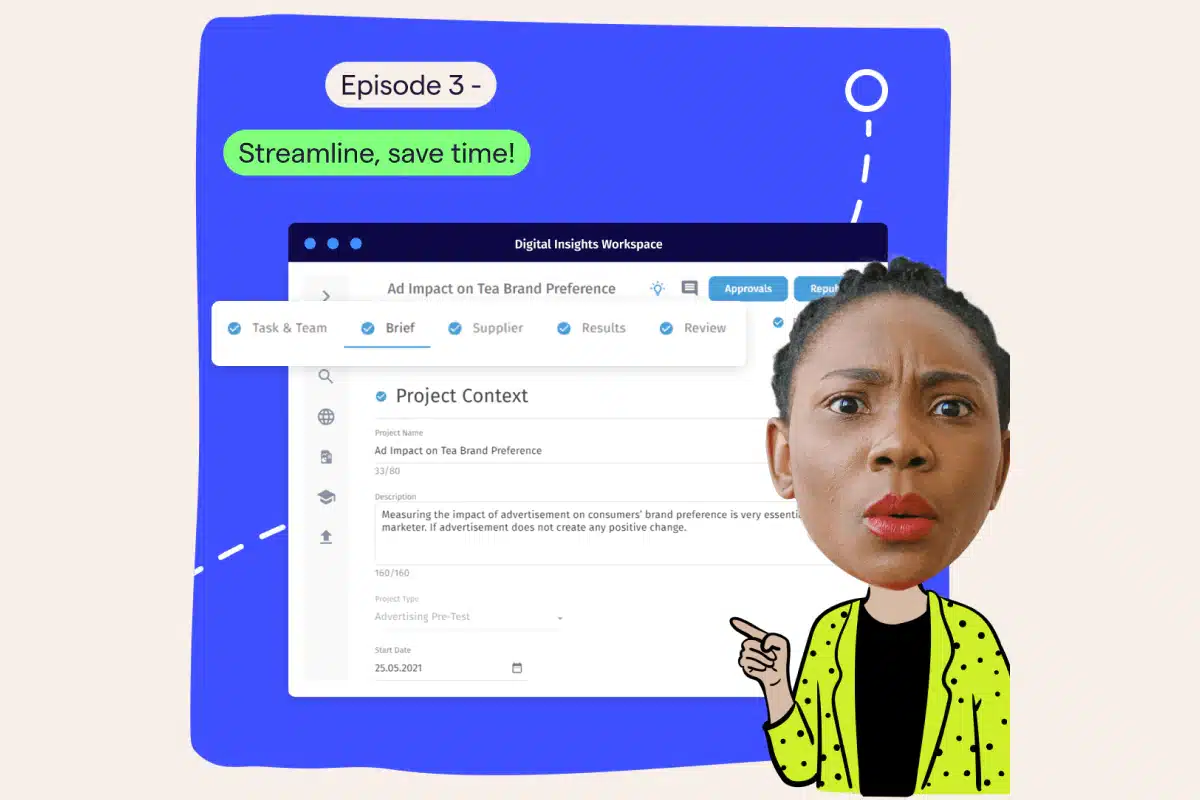
Welcome, everyone, and thanks for tuning in to episode four of our insights boot camp, the last part of this webinar series. If you want to learn tips and tricks on bringing your insights to the next level, you are joining the right session. My name is Tanja Kratz de Suarez. I’m a product marketing manager at Magalogic and your host for today’s session. Alongside me is my colleague and expert in customer onboarding, Sabrina Buschman, who will guide us through a comprehensive platform demonstration later in the session. Before we dive in, let’s cover a few administrative details. Our sessions are being recorded, and a link will be shared in a follow-up email allowing you to access and stream them at your own time. Also, we’ve reserved, some time for questions towards the end of our presentation, so please don’t hesitate to share your questions in the q and a chat box located here on your on twenty four screen. We have covered an exciting curriculum over the last four weeks. In each episode, we have explored a different use case, all of which are probably very well known to you as insights and intelligent professionals. In the first half of this webinar series, we dove deep into how insights and intelligent reaches the business to inform actions. In how to tell powerful stories, we zoomed in on insight storytelling and how you can become a storyteller using MarketLogic’s engaging and easy to use knowledge zones. In episode two, deliver insights faster, we showed you how to deliver fast paced market and competitive intelligence at scale. In streamlined safe time, episode three, we have to insights development, how to automate workflows, templatize best practices, and make use of existing knowledge to run efficient market research. In case you haven’t had the chance to join us in these previous episodes, I suggest viewing the recordings. The email containing the links to watch all past episodes should already be in your inbox. So today, you will learn how to ensure full compliance with internal and external requirements while efficiently managing your research program. Before we jump right into today’s topic, I wanna say a few words on who we are. MarketLogic is the industry leading provider of insights management solutions aiming to guide companies towards becoming insights driven. Leveraging AI technology and tailored for insights engagement, our platform supports consumer centric businesses in providing their leaders with reliable insights at scale, enabling them to make more impactful decisions. With a track record spanning over fifteen years, we have enabled numerous consumer oriented brands worldwide to transition into enterprises that truly put insights at the core of their business operations. Well known market leaders like Tesco, Unilever, or Vodafone rely on market logic to fuel their daily innovation efforts. Today’s episode is especially relevant for highly regulated industries like pharmaceuticals, health care, but also financial services. We know that compliance is not a compromise and that market research is a huge investment that needs to be managed well to minimize financial risks. But even if you’re not working in these industries, you will find many of the tools we’re about to show you very helpful as truly addresses many of the pain points of insights managers and leaders alike. But before we jump to the solution, let’s outline some of the challenges and pain points you may experience. Complex regulatory frameworks must be managed on a project level by insights managers, leading to a numerous of additional administrative tasks involving many different stakeholders. The balance between a guillotine insights and efficiency with compliance. Insights teams are under pressure having to deliver research projects more quickly at lower costs while at the same time meeting high compliance requirements and fragmented systems and processes leading to a number of issues, many manual steps that increase the risk of inaccuracy, inconsistency, and noncompliance, time wasted aligning stakeholders and chasing deliverables, and manual regulatory reporting and collecting information that’s scattered across multiple systems. So what can organizations do to overcome these challenges? A unified and digitalized solution supporting the entire end to end process, replacing the pieced together system. This fosters transparency across all stakeholder groups, removes friction and inefficiencies in the process, and ultimately enables insights team to more effectively manage their projects. Create a single source of truth so that all project information that supports audits are easily and reliably accessible. And automated workflows to enforce and monitor compliance. That includes automated reporting, automated approval processes to really speed up the process and minimize the risk of noncompliance. This is where MarketLogic’s research management toolbox can really support. It provides an end to end solution with a configurable workflow and approval engine. Every step of the process is tailored to the needs of the organization, reflecting ways of working and research practices. Supplier bidding and management, automated regulatory reports, and vendor ratings all empower the insights teams to evaluate, monitor, report, and optimize their work. Now let’s jump into the platform to see hands on how these, research management capabilities work and can really support you in achieving full compliance. Sabrina, over to you. Thank you, Tanja. Hi, everyone. Thanks for joining also from my side. My name is Sabrina. And just for those of you who are joining us for the first time, I work as a customer onboarding specialist here at MarketLogic. Now as you’ve heard from Tanja, today, we will focus on compliance, an important topic for any company, but even more crucial in highly regulated industries. We do have quite a few customers that are big players in the pharma as well as the financial services world, and our platform supports them running research efficiently with one hundred percent compliance is the basis of every single step. I think at this point, it’s safe to say that we understand pharma. And today, I would like to show you an example of that. I’ll show you how research automation with a full audit trail allows your teams to work more quickly, to have full visibility of the whole process, to manage complexity, and essentially to focus on more value added and enjoyable job elements. If you were able to join our last webinar, you have already heard me talk about standardizing workflows, utilizing templates and blueprints to essentially streamline efforts. If this is something that you’re interested in and you weren’t able to attend last week, I can only recommend having a look at the available recording of our episode three. Today, we won’t actually start a research project from scratch, but, instead, we will have a look at a prepared one to focus on aspects of project reminders and notifications, research vendor performance overview, approvals and alignments, the project activity feed, and then finally, automized regulatory reporting. So let’s go ahead and get started with that. Now looking at my Joe page already brings me here to our first topic, project reminders and notifications. Because what we see here is a to do list, my to do list, that includes different tasks and milestones of different research projects that have been assigned to me. And this, yeah, visual reminder of what I have to do by when Joe only exists right on the Joe page whenever I log in, but this is also something that is, of course, part of the respective project as well as can be set up as email notifications. So for example, when a due date is approaching or when milestone has been missed by a couple of days, you can be notified in your normal inbox to to remind you essentially to complete the task at hand. Notifications can be set up at different stages of a project. You can also think of, for example, if you if you have an approval process, all the approvals have already been given. You can then set up an automatic notification to the external research vendor so that they know that they can start with the field work. Now as I said, I have prepared a project for us today. So let me just click on one of my tasks over here that will take us right into the project. And you can see the normal project set up. We talked more about this last week. We do have the different stages here at the top. And, again, under task and team, we have all of our goals, and each individual goal here can be defined with a start date and end date, a responsible person. You can set up notifications and reminders for this that go out automatically. And then whenever someone has completed a task, they can just check it off and move on. I’m now jumping to the next step, the project brief. And over here, I want to touch upon two different topics. One, just super high level approvals, approval flow, but we will come back to those as well. And then secondly, I wanted to, yeah, talk a little bit about suppliers, the external research vendors that you might are collaborating with. So let’s start with our approvals because one thing that you will Joe, especially if you already joined last week, is in my project brief, I have different sections. I see the project context, the scope, and then the brief. And you probably have noted that two of those sections are currently locked for editing. The project scope, I cannot make any changes here, and the same is true for the context. Now why is that the case? This is currently locked because I have already requested and received an approval for those areas of the project. And I just wanted to show you that at any point in time, anyone that is part of the project can, of course, have a look at the full approval flow. So here on the left hand side, you can see the different approvals that have been set up for this. And I do just wanna take a moment here to also differentiate between an alignment. So the first one over here, the brief input request, this is what we call an alignment. Essentially, yeah, technically, an optional approval where you can just easily ask people even from the business, your your team, your manager. Ask them for input, for feedback. You do it, and it’s all documented in the right place. Those alignments are option not only for you to request, but also for the for the aligner, so the person that you’re asking for feedback. Here, I can see one person has already reviewed my work. The other hasn’t. I can send a reminder if I want to so that they just receive this email again. The alignments are often set up for the brief, but, of course, especially with today’s topic of compliance, we do have quite a few actual approval set up. You can see here the the ones, that are actually locking my project context, my project scope. This is the multi country safety approval. It’s maybe interesting to note that all of those approvals have been set up with the right person as the approver based on the project type, the region, the category Joe that the the insights manager, the person requesting them, does not even need to think about, oh, who has the authority to green light this? This is all done automatically and can be set up, of course, per approval step for you. And then once an approval has been requested, nothing can be changed anymore. If the approval has been provided, nothing can be changed anymore, at least not in the current setup. If any edits need to be made, of course, that’s possible. But the approval in this case would need to be canceled so that everything is properly documented. Then once the cancellation went through, you are able to edit the locked previously locked sections again. So here now I can just make a little edit, and then you can rerequest the approval as I’m showing you now. Everything is, of course, properly documented, full transparency, full visibility for the approvers as well as the requesters. If you click on this history icon, you will see exactly what has happened, when, what’s initiated, by whom, and so on and so forth. So this was a first excursion into our approval process and the world of alignments and approvals. I also said I briefly wanted to talk about external vendors. So the way that, in this case, the project brief, but, of course, any project of yours is set up, completely depends on your working reality, your ways of working, your research processes. And I just wanted to showcase briefly that we can set up fields that are essentially triggers for additional fields, even additional tabs Joe that a a even a complex project just only have as many fields and sections as it needs. And if something is not necessary, we won’t even ask people about it. They won’t even see the fields. It should be really as easy as, yeah, somehow possible. Here, we have a question as part of the brief whether this is an internal research. If I now say this is an external piece of research and it does involve an external vendor, then the system will actually add in an additional tab here at the top for us. So this is what I was, yeah, just going to show you about, dependencies and triggering extra fields, extra steps, extra tabs if necessary, if needed Joe that those people who do not need to follow them are not bothered and don’t have a cluttered up experience at all. So let’s go ahead and have a look at the new tab that just appeared for us, the supplier tab. And in our test project for today’s demonstration, this has been set up to essentially just, yeah, ask the insights managers about different parameters of the project, the budget, whether supplier bidding is needed or not. So all of this, of course, could be configured and set up then for your working reality. What I did wanna show you here is that for the supplier selection, we can set this up in a way that some of the suppliers are on roster, meaning those are the preferred suppliers that have been collaborating with your organization frequently, successfully in the past, that have good ratings, and that for this specific project type, the methodology, the region are already Rini it. So there is no additional approval necessary. If I go here to my on roster suppliers, this, of course, helps your insights managers to make the best choices, first of all, to always go with the best agency for the project at hand. And it saves time because no additional steps, no additional involvement of anyone to review. Maybe a local agency is required here. And then we’re just following here the normal, yeah, supplier collaboration. We talked about this more in detail in last week’s episode. So definitely do go back if you’re interested in in learning more. I will already move on here to tab number four, the research planning stage of my project. Now what you would see in here is, of course, again, a lot of fields that have been set up to to essentially ask people whether specific information is necessary, is is needed, is already there, has been provided. And here, we are talking again about field dependencies. So an answer to a question triggers an additional field, an additional step or a tab to really make sure that everything is in order, documented properly, but we’re not including unnecessary steps where they may be not needed. I’m coming down here now. I have already filled out a lot of those fields. But, again, I wanted to highlight, of course, this is all customizable. We will set this up for for your business reality and include the questions that are crucial for your business to have documented somewhere. So here, we’re talking a lot about different reviews that that might be required. And again, the answers that are given to the questions, they trigger additional fields, additional placeholders where you can upload documents, where you can elaborate on the details here Joe that everything is always stored completely in the same place here within your project container. Once all of this is filled in here, I’m just gonna keep going. You might find an approval that has not been approved yet. So this is the one that I just left for the demonstration purpose of today. I wanted to show you what this looks like. Again, depending on the project specifications, the system knows exactly whom to choose as an approver. I’m just going to go ahead and request this. Joe what I can do here is I can enter the person who is the initiator of the research project. In our case, that is my colleague, Tanja, over here. The, some of the approvers I can choose myself, and others have already been selected based on country, region, category, project type, so that that your insights managers, they don’t need to think about this. They don’t need to think who’s the right person as I mentioned before. I’ll just send off this automatically generated email here. You can see all of the approvals have been requested. They’re dependent on one another. So the moment that Tanja here, provides her approval, the next person is notified. I’m an admin user, so I’m just gonna show you what this looks like. I can force approve, and this now means it’s Ernest’s turn over here. And then once he has provided his approval, it goes on to the next person. Now, while we have already talked about how approvers lock certain sections for editing, it is, of course, also crucial for the approvers to understand exactly, have they been made any changes. So I just wanted to make you aware that for each field and for each document, we do have a history icon. So if there was any previous input given, you can always see exactly here locked with a date and an ownership stamp, who did what when, just to to have this completely available, of course. Also for the people reviewing your content, your work, and then greenlighting it in the end. Exactly. Now from here, I would now like to move on to our next stage, the project planning and compliance safety. Again, a lot of the approvals here have already been provided. But this is all about just again showing you that we do have the option to add any mandatory element that you have, that your company has. In here, we can have the drop down questions. Depending on the answers, You’re gonna find, a place to upload documents or to to just, yeah, provide more details as well. So this is all designed to really have everything, all the documentation in one place and to to save time to make it available to whoever needs it very quickly whenever it is actually needed. And I’m moving on to the final deliverables over here. Joe this is information that the suppliers actually upload. If you have the supplier collaboration, of course, it would be possible for you to do the same. Just wanted to remind you of the the change logs, the full transparency, full visibility at any stage. If we see, for example, that we have a version two of a full report, we can just have a look at the history and again we’ll see exactly what happened when, by whom. We can look at all of the documentation provided also in the past. Only the most recent one, of course, would be published if this goes now into the database. Exactly. We already talked about the timestamps here. So this is something that we we like to include at several stages just so that we have an official acknowledgment that all of the proper documents have been submitted and provided and so on and so forth. From here, we are now essentially moving on to publishing. And in publishing, we are rating our supplier, the external research vendor. This is actually quite important and quite, yeah, also a a a helpful feature, I will say, because not only can the insights managers provide their rating here, I’ll just give them a few stars. If something is exceptionally good or not so good, you can add in a comment, of course, as well. But not only do we fill this in as insights managers to learn from one another, but and now I’m jumping a little bit. Please bear with me. There’s also a supplier directory. If you follow me here to the top of the screen, what you will see is that I have my full overview of the vendor performance essentially. If you think about people, let’s say, from procurement, for example, who sit down every quarter, maybe once a year with the vendors, they can actually go ahead and just download all of this information. So the supplier performance export, including the comments, the star rating, how many projects have they worked on, how much money have we spent with them. And they have this readily available for any, yeah, upcoming review meeting that might, needs to be scheduled in the future. So I would say this is also an important part here the the assessment before of course publishing everything. And talking about publishing of course we always say sharing is caring but of course, it is possible, especially, of course, in those in those worlds where we talk about the pharma industry or the the financial services to publish content only to a very small group of people. This is at any point in time, of course, changeable. If if maybe information can be shared down the line in a year from now, this can always be adapted. But for the moment, maybe we just wanna keep it in the small group. I’m gonna go ahead and publish this, and then it’s available only to the people that I have included here in the in the the the target audience. Now, there are two more things that I wanted to talk about today, and I will open up the project again because, yeah, those are actually quite important when we talk about compliance and talk about being able to yeah. To to essentially prove everything that was done at the speed of light essentially. You have two options to do that. The first one would be our activities feed. If you click here on the little clock icon, you will see all the actions that have been triggered with timestamps with the owner who did what when. And, of course, those can also be downloaded. So upon request, download it, send it to whomever needs it, and they will have a full record of everything that is part of the project here immediately available. The second thing that I wanted to mention here, I’m not gonna show you, but I at least wanted to talk about it. This is all about automated regulatory reporting. If you think about the pharma, Covaligence, for example, what we can do is we can set up automated automated reports that are sent to the right stakeholders in a cadence that you decide on. So if it’s every quarter, every half a year, we can set up different reports with key information of the project. And then maybe that’s sent to, I don’t know, the the safety team, the IT team, the compliance team. And everything happens automated in the background. No one needs to remember and think of this. No one needs to download and send it. So those, yeah, optimized regulatory reports can, of course, also be set up by the MarketLogic team for you. Yes. And this is everything that I wanted to show you today. I I think when we talk about key takeaways here, I think it’s fair to say that compliance is a given. We’re we’re we’re not talking about this. It’s a basic requirement. I just wanted to show you some some ways and some features that we have in place to support you and other organizations to achieve this at any point in time. And back to you, Tanja. Thanks, Sabrina, for this comprehensive, platform demo. Now let me summarize the key takeaways. It is possible to run agile research that is in line with rigid compliance requirements. How? A unified system that allows for documentation and tracking of all project deliverables and information to satisfy any reporting needs. You can set up a governance system with an approval engine that is highly flexible and removes barriers to deliver insights projects faster. Involve your key internal stakeholders. Make sure that, for example, your safety or legal or ethic teams actively participate in the process. There are several ways to do that, you know, as approvers, where they’re acting as gatekeepers. They have access to all the relevant information right in the tool that’s informing their decisions, or they can be invited to provide feedback and alignment and to really move that collaboration into one place. Okay. They can just have access to the to the project inbox, for full transparency on what research activities, are available across the business. Or maybe they’re involved as internal auditors who require information for regulatory reporting. Just make sure you gather the requirements and you actively include them. It also brings me to the last point, automated reporting. Now this is addressing a big frustration among insights teams that have to adhere to high, reporting requirements. So with a unified solution that covers the end to end process, where all information already available to automate reporting based on the needs of the internal but also external teams. Now we have a minute for questions. Let me take a look at the chat box here. Are the research projects connected to the knowledge management system? That’s a good question, because we have actually not seen that in the demo, and the answer is yes. The idea really is that all the fresh insights that come out of these, you know, research projects are being fed seamlessly into the knowledge base so that the wider business has business has instant access to them. And since we’re running a little bit out of time, let me just give you a quick outlook of what we have in the pipeline to support, the research process. In the future, you will be able to run desk research within the tool supported by the AI summarizing your existing knowledge from internal and external sources. So this is going to be a big time saver in understanding what’s already available to help in forming your research agenda. Already available today is our AI assistant for trusted insights called DeepSights. Simply ask a natural language question and get immediate answers that are based on your knowledge and data. That means you get answers that you can trust, so no AI hallucination that you may know from other public AI tools. If you’re interested and you wanna explore it yourself, just use the barcode here on the slide. Now that concludes our boot camp series. Thank you so much for joining. I hope you enjoyed the sessions and got something out of it. If you have any follow-up questions or you’re ready for a personal demonstration, please do reach out. You see our contact details here on the slide. Looking forward to seeing you on any of our upcoming events. Thanks again for joining, and, goodbye.
High regulatory demands and extreme scrutiny of market research are the reality for many industries such as Healthcare & Pharmaceuticals. Learn to leverage Market Logic’s research capabilities to run end-to-end research. Avoid duplicative efforts and standardize best practices, with foolproof compliance.
Stream this episode to learn:
- Run approval engine to support achieving 100% compliance.
- Improve your insights and procurement team’s efficiency by gathering quantitative and qualitative feedback on outside vendor performance.
- Provide visibility on business-wide research activities with automated regulatory reports.